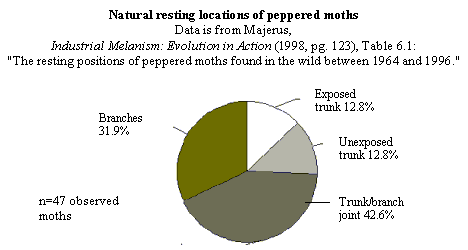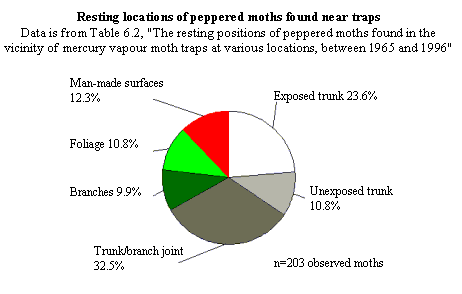First, several of Wells' worst distortions must be
dealt with directly.
-
The natural resting locations of peppered moths -- Majerus' data.
On page 148, Wells discusses the natural resting places of peppered moths,
under the heading "Peppered moths don't rest on tree trunks." But they do,
at least sometimes. Here are the relevant datasets, which Wells does not
quote or cite for his readers:
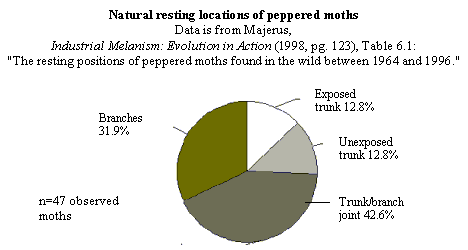
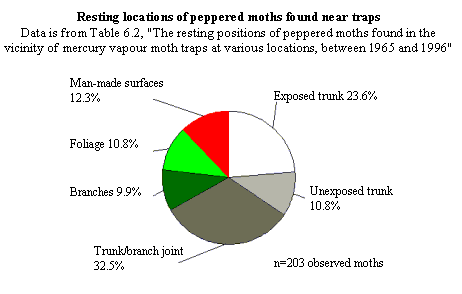
For further discussion, see below and endnote 3.
-
Peppered moth photographs, staged and otherwise. Wells raises a
fantastic stink about the fact that the photographs of peppered moths in
textbooks, showing light-colored typicals next to dark-colored melanics on
differing backgrounds, are staged. But the point of such photos is not
to prove the truth of the 'classic' story, it is to illustrate the relative
crypsis of moth morphs on different backgrounds. Those who feel that
their innocent faith in insect photography has been betrayed should consider
the fact that most photos of insects in textbooks are probably staged; insects
are, after all, small and difficult to photograph. The facts that peppered
moths are sparsely distributed and, well, camouflaged also make them
difficult to photograph.
But as it turns out, the differences between staged and unstaged photos are
minimal. Readers who wish to see unstaged photos of peppered moths are advised
to look up Majerus' Industrial Melanism. Majerus says that all of the
peppered moth photos taken by him in the book are unstaged. Readers should
consult the figures which are listed below. It may be possible to get
permission to include the photos, but until then descriptions shall have to
suffice.
(For those with foggy memories of their texbooks, English peppered moths
come in three general phenotypic categories: typica, the pale, original
'peppered' form of the moth; carbonaria, the almost black melanic form;
and insularia, which includes a range of intermediate-colored
moths.)
-
Figure 6.1 (a), p. 118. Black-and-white photo, edges blurred. A
rather dark (almost black) insularia moth, resting apparently on a
tree trunk (bark fills the background). The moth is slightly darker than the
background.
-
Figure 6.1 (b), p. 118. Black-and-white photo, middle of moth
slightly blurred. A light form of insularia (still more heavily
peppered than a typica), resting on a thick tree branch (branch width
is about 3/4 that of the moth).
-
Figure 6.3, p. 122. Black-and-white photo, middle of moth slightly
blurred. A typica hanging underneath a hazel twig.
-
Plate 3, between pp. 146-147, has colored photos. Six photos are
shown (the first five are Majerus'), and the captions are quoted, with my
comments in brackets.
-
(a) "Typica and carbonaria forms of the peppered
moth on an [sic] horizontal birch branch." [This situation, with two moths
close enough together to photograph at once, is very rare, basically only
occurring if two moths are meeting to mate.]
-
(b) "A pair of peppered moths on a twig at dawn. The
carbonaria male is much less conspicuous than the typica
female." [The carbonaria moth is quite blurry.]
-
(c) "A carbonaria peppered moth in shadow under a
horizontal branch, showing how this positioning may reduce the likelihood
of detection." [The moth is being viewed head-on and is indeed difficult
to see.]
-
(d) "Typical form of the peppered moth at rest during the day in
hazel foliage." [Head-on view, the moth is hanging underneath a thick
twig.]
-
(e) "An intermediate, insularia form, of the peppered
moth." [A 'classic' view, the moth is well-matched to its background,
which is apparently tree bark.]
-
(f) "The non-melanic form of the peppered moth from North
America, Biston betularia cognataria (courtesy of Professor Bruce
Grant)." [A 'classic' view, the moth is well-matched to its background,
which is a lichen-covered surface.]
It should be noted that Majerus is concerned to show his readers aspects of
the peppered moth story that they do not get in textbooks; thus the focus on
insularia forms and on moths in branches (Majerus is a proponent of the
view that peppered moths most commonly -- but not entirely or even
almost entirely -- rest on the underside of branches and thick twigs in the
forest canopy). Even so, there are several photos that show peppered moths, on
tree trunks, on more-or-less matching backgrounds. And guess what? These
photos look no different than 'staged' photos of moths on tree-trunks.
The most 'staged' aspect about a 'staged' photo is that two differing moth
forms are shown side-by-side, but Majerus' first two photos from Plate 3
indicate that even this is not impossible. So the entire photo issue is a
mountain made of a molehill.
It should also be noted that several (four) of these unstaged photos have
some (minor but noticeable) degree of blurring (e.g., part of the moth will be
out of focus). Insects in the wild do annoying things like move and fly away,
and are often encountered in poor-light conditions, resulting in
less-than-perfect photos. As scientific documentation of observations this is
unimportant, but flawed photographs are exactly the kind of thing that are
avoided in textbooks, and this is precisely why staging insect photos is a
common practice for textbooks (as well as things like nature
shows).
Summary of Wells' treatment of moth resting places. To review, Wells'
primary objection to the peppered moth story was this:
Most introductory textbooks now illustrate this classical story of natural
selection with photographs of the two varieties of peppered moth resting on
light- and dark-colored tree trunks. (Figure 7-1) What the textbooks don't
explain, however, is that biologists have known since the 1980's that the
classical story has some serious flaws. The most serious is that peppered
moths in the wild don't even rest on tree trunks. The textbook photographs, it
turns out, have been staged. (Icons, p. 138)
[Figure 7-1 is on Icons, p. 139; these are drawings by
Icons illustrator Jody F. Sjogren; the source photo, if there is one, is
not cited. Confusingly, the caption for the figure is not on page 139 but
overleaf on page 140. These are not encouraging signs in a book purporting to
critique textbooks.]
The discussion thus far has shown that Wells' "most serious objection" to the
peppered moth story is completely baseless: first, peppered moths do in fact
rest on tree trunks (a significant portion of the time although not the majority
of the time, according to Majerus' data). Second, textbook photos are used to
show relative crypsis of moth morphs, not to prove that peppered moths
always rest in one section of the trees. And third, Majerus himself has taken
unstaged photos of peppered moths on matching tree trunk backgrounds, and these
are not significantly different than staged photos; this eviscerates whatever
vestige of a point Wells thinks that he has.
-
What are the implications if moths rest most often underneath
branches? Leaving aside Wells' frantic attempt to create a problem where
none exists, the relevance of moth resting locations for the 'classic story'
(natural selection by bird predation) deserves some consideration. Majerus'
considered opinion is that peppered moths rest more commonly underneath
branches than was previously appreciated, and that if this is true then some
quantitative estimates of selection coefficients may need to be adjusted.
However, he is quite clear that the basic qualitative conclusions of
Kettlewell (that differential bird predation of moth morphs on changing
backgrounds is the selective force) do not need to be changed. As Majerus
notes, crypsis is still important for moths in tree branches. He even comments
directly on this with two of his photos (Plate 3, photos (b) and
(c)). And of course, birds are known to (a) fly and (b) feed in forest
canopies, so it is very difficult to see why resting on trunks vs. branches
would change bird predation in any radical way.
The scientific literature.
Having dealt with Wells' "most serious
objection," let us turn to Wells' use of the scientific literature. The primary
problem is that Wells gives inordinate weight to a few scattered review papers,
by biologists who are not major peppered moth researchers [3], that
question the standard view (that bird predation on different colored moths on
differently polluted backgrounds caused the darkening of moth populations as
pollution increased, and that as pollution decreased this process worked in the
opposite direction). Their criticisms have been answered by peppered moth
researchers (Grant,
1999; Cook,
2000; Grant and
Clarke, 2000; Majerus, 2000).
And, as pointed out in the introduction, since Wells bases his argument on the
idea that the experts are disowning the 'icons' in their respective fields,
Wells is falsified if those experts contradict him.
-
Bruce Grant's review of Wells.
American peppered moth researcher
Bruce Grant has written many papers on Biston, and has documented the
parallel rise and fall in melanic forms of the North American subspecies of
the peppered moth. See Grant's webpage for
listed articles. Dr. Grant has kindly given permission to have his comments on
the peppered moth chapter of Icons quoted in this article.
To put them in context, the material quoted below is a copy of the
correspondence between Grant and a professional colleague who had requested
Grant's views on Wells' chapter, originally written February 7, 2001.
Subject: Wells' Chapter on Peppered Moths
Wells' Chapter 7 is pretty similar to his earlier ms. "Second thoughts
about peppered moths" that he posted on the web, and published in abridged
form in The Scientist. I sent you my comments about that version
about two weeks ago. My general reaction to this latest version is about the
same. He distorts the picture, but unfortunately he is probably pretty
convincing to people who really don't know the primary literature in this
field. He uses two tactics. One is the selective omission of relevant work.
The other is to scramble together separate points so doubts about one carry
over to the other. Basically, he is dishonest.
He immediately launches the claim "that peppered moths in the wild don't
even rest on tree trunks" (p. 138). This is just plain wrong! Of course they
rest on tree trunks, but it's not their exclusive resting site. He quotes
Cyril Clarke's lack of success in finding the moths in natural settings, but
he omits mentioning Majerus' data which reports just where on trees (exposed
trunks, unexposed trunks, trunk/branch joints, branches) Majerus has found
moths over his 34 years of looking for them. Of the 47 moths he located away
from moth traps, 12 were on trunks (that's >25%). Of the 203 he found in
the vicinities of traps, 70 were on trunks (that's 34%). Based on his
observations, Majerus argued that the most common resting site appears to be
at the trunk/branch juncture. What is clear from his data is that they sit
all over the trees, INCLUDING the trunks. So what? Kettlewell's
complementary experiments in polluted and unpolluted woods compared the
relative success of different colored moths on the same parts of trees in
different areas, not different parts of trees in the same area. It is true
that the photos showing the moths on trunks are posed (just like practically
all wildlife pictures of insects are) but they are not fakes. No one who
reads Kettlewell's paper in which the original photos appeared would get the
impression from the text that these were anything but posed pictures. He was
attempting to compare the differences in conspicuousness of the pale and
dark moths on different backgrounds. Nobody thought he encountered those
moths like that in the wild. At their normal densities, you'd be hard
pressed ever to find two together unless they were copulating. I have always
made a point of stating in photo captions that the moths are posed, and I
think textbook writers have been careless about this. But they are not
frauds.
On the subject of lichens, no one has questioned their importance more
than I have. But what does Wells do with this? He quotes me, but he doesn't
include what else I said has happened on the Wirral (p. 147) with respect to
the tremendous expansion of birch stands since the enactment of the
smokeless zones. Kettlewell, too, argued that peppered moths are well
concealed on birch bark (even without lichens). Wells continues (p. 148) to
quote my reservations about lichens in Michigan, but, again, he omits any
reference to the data I presented in that paper showing the decline, not
only in SO2, but in atmospheric particles (soot) which has been established
as a factor altering reflectance from the surface of tree bark. So, while I
have questioned the importance of lichens, I have not taken this as evidence
that crypsis is unimportant. Wells omits this entirely.
Wells continues to bring up the same old arguments about mysterious other
factors (yet to be identified) that account for the persistence of typicals
in polluted regions, and the presence of melanics in unpolluted locales. He
cites papers written back in the 70s about these puzzles. He omits
discussing in any sophisticated way the role of migration other than to say
"Theoretical models could account for the discrepancies only by invoking
migration...." (p.146), as if in desperation we are forced to grasp at
straws. Of course migration is important. Majerus actually reviews this
point fairly well by comparing the smoothness of clines in melanism between
species that are highly mobile (as is Biston), and species that are
relatively sedentary. Instead of showing his meaningless map of the UK (Fig.
7-2) to illustrate what he regards as anomalies in the distribution of
melanism and lichens, why doesn't he show the before and after comparison
from the national surveys by Kettlewell in 1956, and the survey by Grant
et al. in 1996. (If you'd like, I can send you a jpg file of the maps
I mean.)
Wells also inappropriately uses thermal melanism in ladybirds to suggest,
that while no one has shown this in peppered moths (p. 152), industrial
melanism can have other causes besides predation. It's not just that there
is no evidence for thermal melanism in peppered moths, there is evidence
AGAINST thermal melanism based on the geographic incidence of melanism in
the UK, the USA, and Europe. There are no latitudinal clines, and no
altitudinal clines as one might expect with thermal melanism. Wells knows
this, if he actually read my papers. (He cites them, so I should assume he
read them.) He also raises the question of larval tolerance to pollutants.
There is no evidence for this, either. I have a paper out on this point, but
in fairness to Wells, it came out just this past year.
Wells clouds discussions with irrelevancies. For examples he brings up
Heslop Harrison (p. 141 and again on p. 151) and the question of phenotypic
induction. Wells makes it sound as if most biologists discount induction
based on their belief in natural selection (as if it were a popular
religious question). The evidence for the Mendelian inheritance of melanism
in peppered moths has nothing to do with evolutionary theory; it is based on
old fashioned crosses involving over 12 thousand progeny from 83 broods. The
Mendelian basis for this character in this species is as well established as
is any character in any species. Wells doesn't mention this, yet he cites my
review paper where I do bring this up in my criticism of Sargent et
al. Induction has nothing to do with industrial melanism, and Wells
knows it. Again, selective omissions on the part of Wells.
On page 151 Wells claims Kettlewell's evidence has been impeached. This
is nonsense. It has not. But I have argued, that even if it were entirely
thrown out, the evidence for natural selection comes from the changes in the
percentages of pale and melanic moths. It is this record of change in allele
frequency over time that is unimpeachable. It is a massive record by any
standard. (I can send a jpg file with graphs, if you'd like.) I have pointed
out, and he quotes me, that no force known to science can account for these
changes except for natural selection. Yet, he scrambles the ingredients
here. He claims (top of p. 153) "...it is clear that the compelling evidence
for natural selection that biologists once thought they had in peppered
moths no longer exists." Of course the evidence for natural selection
exists! That evidence is overwhelming. Wells, by attempting to discredit
Ketttlewell's experiments about predation (and clearly there are things
wrong with Kettlewell's experiments) doesn't stop at saying we can't be
altogether sure about bird predation because of problems with Kettlewell's
experiments. No. He says, instead, that the evidence for natural selection
no longer exists. This is just plain wrong. He cannot support this sweeping
statement, but he spins it into his conclusion by building a case against
Kettlewell. This is what I mean about his tactic of scrambling arguments. He
wields non sequiturs relentlessly.
I hope this is helpful to you in your review.
Bruce Grant, Professor of Biology, College of William & Mary.
February 2001
M.E.N. Majerus' review of Wells
-
Majerus, who as we have noted wrote
the (most recent) book on peppered moths and industrial melanism (Majerus, 1998),
wrote a detailed critique of Wells' earlier treatment of peppered moths in
1999. Here is Majerus' opinion of Wells' treatment of peppered moths (in part;
see the links below for the entire correspondence online):
[...]
Evidence of selective predation in the peppered moth is not lacking. It
is just not provided in the quick text book descriptions of the peppered
moth. How can it be. I have read some 500 papers on melanism in the
Lepidoptera. In total, these papers probably amount to about 8000 pages, and
the story is condensed into a few paragraphs in most textbooks for schools.
Even in my own book, I could only give a review of the case covering about
60 pages including illustrations.
The older hypothesis that melanism was induced by pollutants was
discredited because Heslop [Harrison's] experiments lacked appropriate
controls, and his results could not be replicated, despite several attempts.
Furthermore, the levels of mutagenesis that he recorded are several times
higher than those produced by doses of radiation that induce complete
sterility in fruit flies (see E.B. Ford (1964) Ecological Genetics for full
critical review).
Finally, I agree with Dr Wells that photographs of two peppered moths
staged on backgrounds for effect should say they have been done purely for
illustrative purposes. I have many times, in undergraduate lectures, pointed
out that photographs of the type that appear in so many text books are
faked. However, I would point out that none of the photographs of live
peppered moths taken by myself, which appear in the book were staged. All
show peppered moths where they were found in the wild.
End-note: It is difficult to have an informed discussion of a complicated
ecological system with those who have little or no experience of the system.
My advice to anyone who wishes to obtain a fully objective view of this case
is to a) read the primary papers that I based my review upon, and any other
relevant papers, and b) gain some experience of this moth and its habits in
the wild. Of all the people I know, including both amateur and professional
entomologists who have experience of this moth, I know of none who doubts
that differential bird predation is of primary importance in the spread and
decline of melanism in the peppered moth.
I hope that this is some use to you, Donald, and that it encourages more
people to look at the case of the peppered moth with an open mind. If it can
help interest a few more people in moths and butterflies, that is all to the
good.
Best wishes, and Happy Easter.
Mike Majerus
(Majerus, 1999)
-
The above was posted to the Calvin listserv in Donald Frack's detailed
investigation of Wells' claims (e.g. Frank's Peppered Moths -- Round 2).
In his response, Wells freaks out and calls Majerus (who had previously been
Wells' main source for his "peppered myth" claims) a fraud:
BUT EVERYONE, INCLUDING MAJERUS, HAS KNOWN SINCE THE 1980'S THAT PEPPERED
MOTHS DO NOT REST ON TREE TRUNKS IN THE WILD. This means that every time
those staged photographs have been re-published since the 1980's constitutes
a case of deliberate scientific fraud. Michael Majerus is being dishonest,
and textbook-writers are lying to biology students. The behavior of these
people is downright scandalous.
I know what I'm talking about. I spent much of last summer reading the
primary literature (email me if you want the references). Frankly, I was
shocked by what I found -- not only that the evidence for the moths' true
resting-places has been known since the 1980's, but also that people like
Majerus and Miller continue to deceive the public.
Fraud is fraud. It's time to tell it like it is. (caps original, Chadwick's Peppered Moths Again)
Frack says of this,
Note the complete irony of the capitalized sentence. Majerus is the
foremost proponent (in the literature I've seen) of the idea that the moths
most commonly rest higher in the trees. His data are the only ones I have
seen cited as evidence of [what] happens "in the wild." Majerus is attacked
as "dishonest" and "text-book writers are lying to biology students", their
behavior is "scandalous." [...] If Wells is right, he hasn't demonstrated it
here. He attacks both Michael Majerus and Bruce Grant. If Grant's frequent
co-authors, such as Cyril Clarke, are added to the ridicule list (and I
don't know why they wouldn't be), then Wells is well on his way to rejecting
all the well-known researchers on this subject. An awesome, and, at face
value, an incredibly arrogant, claim.
-
There is much more where this came from, and unfortunately there is not yet
a comprehensive web source covering Wells' abuse of peppered moths, so you
have to do some digging. Some good places to start are these links:
-
Bruce Grant's review article: Fine Tunning the Peppered Moth Paradigm
-
Don
Lindsay's archive (links to various letters to newspapers from
Grant and others, protesting Wells' characterizations of their work)
-
NMSR/Wells dialog
-
Wells scored a minor coup when a toned-down version of his essay "Second Thoughts
about Peppered Moths" was published in The Scientist
(13(11), p. 13, May 24, 1999). A longer, unedited version is
here.
-
The 1999 Wells/Frack posts on the Calvin listserv go roughly in this
order (these really should be reformatted and archived somewhere):
-
Frack, "Peppered Moths - in black and white (part 1 of 2)
-
Frack, "Peppered Moths - in black and white (part 2 of 2)
-
Wells, quoted in "Peppered moths again"
-
Frack, "RE: Peppered Moths again"
-
Frack, "Peppered Moths - round 2 (part 1 of 2)"
-
Frack, "Peppered Moths - round 2 (part 2 of 2)"
-
Frack, "Peppered moths, round 3"
-
Frack, "Peppered moths and Creationists"
-
Wells, "My last word"
-
Frack, "RE: My last word"
Of moths and maps
So, the experts disagree with Wells and
furthermore identify just the kinds of deceptive tactics that I have been
talking about. A further instance is Wells' Figure 7-2 (p. 145), a map of
England with four locations marked with letters representing "Discrepancies in
peppered moth distribution." This map deserves enshrinement as exhibit A in
geographer Mark
Monmonier's (1996) book How to Lie with Maps. This is basically
what Wells' map looks like:
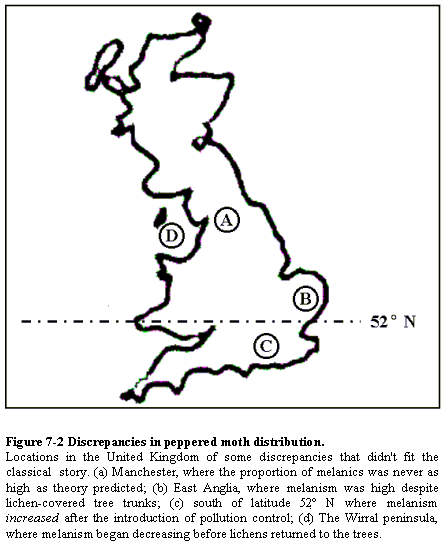
(After Wells,
Icons, p. 145, Figure 7-2. The exact caption is quoted below the
figure. My rendition of the border of Great Britain is very crude, being based
on the first graphic I could find on the internet, but apart from that Wells'
figure is accurately represented. A figure similar to that in Icons can
be found in Wells'
unedited moth essay, here.)
What Wells did here was dig through the literature and find a few instances
where he could find some weak excuse for an observation 'contradicting' what
was expected. However, if one inspects real maps by real moth researchers, one
finds that the geographic pattern is actually a good match with expectations.
Here are the maps that Bruce Grant mentioned in the above-quoted review:
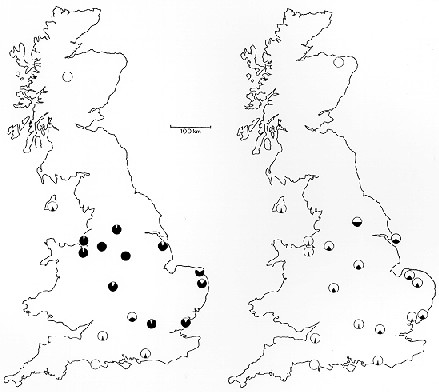
Grant's comments on these maps: "The maps show a before-after comparison of
the geographic distribution of melanic phenotypes in peppered moth populations
in Britain based on Kettlewell's 1956 survey (left map) and that conducted
40-years later (1996) by my colleagues and me (right map). The black segments
of the pie charts indicate the percentage of melanics at the various
locations. Clearly melanism has declined everywhere it was once common."
(Grant, personal communication, February 11, 2002)
The source publication for these maps: Grant, B. S., Cook, A. D. ,
Clarke, C. A., and Owen, D. F. 1998. Geographic and temporal variation in the
incidence of melanism in peppered moth populations in America and Britain.
Journal of Heredity 89:465-471.
Wells' map (Figure 7-2 from Icons) is more fraudulent than all of
the textbook moth photos put together.
8. Chapter 8: Darwin's Finches
"Darwin's Finches" are a group of closely-related bird species located on the
Galápagos islands. The finches have been studied ever since Darwin made the
first collection of these species, but the best-known recent work has been
conducted by the husband-and-wife team of Peter and Rosemary Grant and their
collegues and students. See Peter Grant's 1986 book Ecology and Evolution
of Darwin's Finches and subsequent articles by the Grants, and
especially Jonathan Weiner's 1994 book The Beak of the Finch:
A Story of Evolution in Our Time for a popular introduction to their
work. See also the March-April 2002 issue of American Scientist,
"Adaptive Radiation of Darwin's Finches," by Peter and Rosemary Grant. The
abstract and some graphics are online.
Finches not relevant to Darwin? Wells claims that the finches were not
relevant to Darwin's development of evolutionary theory, but Wells is playing
fast and loose with the facts here. Wells writes (p. 160) that the finches "are
not discussed in [Darwin's] diary of the Beagle voyage except for one
passing reference." Wells (p. 162) does include a quote from the two pages that
Darwin spent on the finches in The Voyage of the Beagle (Wells, like
Weiner, calls it Journal of Researches), but neglects the well-known fact
that this quote constitutes the first publicly published hint of
evolutionary theory from Darwin (Weiner, 1994).
Wells also leaves out mention of the prominent figure that Darwin put in
Voyage of the Beagle, showing the differently-adapted beaks of the
finches. You can read the quote yourself in the online edition of Voyage of
the Beagle.
The various legends that have grown up around Darwin and the finches -- similar
to Galileo and the Leaning Tower of Piza -- are well addressed in Jonathan
Weiner's The Beak of the Finch (p.p. 35-36), minus the patronizing
editorializing of Wells. Darwin made the first scientific collection of the
finches, so the label "Darwin's Finches" is entirely appropriate.
-
The origin of the finches. Wells' figure 8-1, showing the 14 species
of Darwin's finches in a radial pattern, is as usual not very helpful to a
reader interested in the truth. Why does Wells not show the finches in their
traditional classification into four genera (or five or six; see Sato et al.,
2001)?
-
Why, for that matter, does Wells never mention the scientific names of the
finches or their division into multiple genera? Perhaps Wells avoids doing
this because it would undermine his whole argument that the Grants' conclusion
that natural selection (and a few other well-known factors, e.g. isolation) is
responsible for the speciation of the finches is making "more of their work
than the evidence warrants." (p. 174) Wells of course makes no attempt to
propose a better explanation, or even give any reason why their conclusion is
wrong. Wells instead focuses on the back-of-the-envelope calculation of Peter
Grant in a 1991 that under directional selection a speciation event could
occur in 200 years, even though Peter Grant acknowledges in that very
publication that he has observed oscillating selection.
-
Wells pretends that the only evidence for natural selection being
responsible for speciation is the Grants' direct observations. But this is
patently false -- Wells brushes off the extensive character displacement
studies by multiple researchers as "indirect" (p. 164); he notes that
non-oscillating climate or a long-term trend change in the future might make
selection directional, but Wells fails to tell his readers that we have
extensive evidence of just this occurring in the past, e.g. ice ages on the
scale of hundreds of thousands of years; and Wells fails to note that the
oscillating prospects of hybrid finches that the Grants have observed is a
result of competition for resources among individuals of species that
have divided up the available resources by specializing on food sources (this
is character displacement).
-
Wells spends most of the chapter on the Grants' selection and hybridization
studies, but he gives barely a hint of The Main Point of the Darwin's Finches,
namely that overwhelming evidence indicates that these several genera evolved
from a single ancestral species, and that they did so in an adaptive
manner.
-
Wells never stops to think of what might happen if a single finch
population were to arrive on the originally finch-free Galápagos islands.
Currently, the various finch species have evolved to specialize on different
resources (different sizes of seeds, etc.). Thus, the divergence of one
species into two is impeded by prexisting finch species occupying other
niches. But this would not be the case for the first finch population to
arrive at the island. Expansion into new niches would be favored because those
niches would be unoccupied.
-
Parenthetically, Wells completely botches his description of El Niño on p.
168. Wells describes El Niño as "a disturbance in the winter weather patterns
caused by unusually warm air over the Pacific Ocean." ... which is incorrect,
as an El Niño event is actually a change in the sea-surface temperature
gradient across the Pacific. Normally winds push water westwards
along the equatorial Pacific, resulting in the upwelling of cool nutrient-rich
water in the eastern Pacific. When the winds weaken, the warm water "sloshes"
(speaking very colloquially) eastward, resulting in a cooler west Pacific and
a warmer east Pacific, resulting in reduced upwelling (and poorer fishing) in
the eastern Pacific and heavy rains in the Galápagos and in southern
California (see NOAA, 2001 for a
more complete description).
-
An excellent treatment of the finch example is given by Weiner's (1994)
The Beak of the Finch. See also:
9. Chapter 9: Four-Winged Fruit Flies
Wells must have been hard up for icons at this point, considering that in the
text he cites (p. 185) exactly one textbook that uses the example, and
mysteriously leaves this "Icon" out of his textbook evaluation (p. 249). Wells
leaves fossil horses and fossil hominids out of this evaluation, also, although
for different reasons as they are ubiquitously cited in textbooks.
-
Wells' brief (pp. 181-182, see also 245-246) discussion of "biochemical
mutations" (all mutations are biochemical, actually) is another in a long line
of IDist attempts to minimize the importance of things like antibiotic
resistance (and the corresponding importance that such examples confer to
evolutionary biology). Reviews in the mainstream literature, however (e.g. Walsh, 2000) are
never so flippant. Walsh (2000) notes
that bacteria have 'learned' how to pump out antibiotic molecules, destroy
them, and evade them, and discusses several options to new develop new
antibiotics, methods of circumventing bacterial resistance, and strategies for
extending antibiotic lifespan through a more rational use of drugs. A good
discussion of this issue is also found in Weiner's (1994) chapter "The
Resistance Movement."
-
Wells tries (on p. 189) to disqualify all of the above evidence for
beneficial mutations by demanding what he calls "morphological
mutations." Wells brushes off all of the examples of rapid morphological
evolution in the literature (e.g. cichlid mouthpart specialization, Hawaiian
Drosophila, and so forth), as well as the utterly ubiquitous evidence
that changes in genes cause changes in morphology, with a single brazen
hand-wave (p. 190) about how the connection between genetic differences and
morphological differences is "assumed". Wells claims that there is support for
his notions among past biologists and in non-American biologists, but all he
can do in the text is cite a postmodern-sounding argument from a historian. In
the notes he cites a smattering of sources, e.g. Brian Goodwin (no supporter
of extra-genetic inheritance as far as I've heard) and a 1990 article raising
the possibility of somatic inheritance, but Wells doesn't even try to build a
case; presumably he is just trying to up the doubt meter a bit. Wells also
does no serious search for the evidence he says he requires, namely genetic
difference leading to beneficial morphological difference; I, however, was
able to find an example in 30 seconds on PubMed, dealing with the mapping of
genetic loci that correlate with the morphological differences between the
flowers of two closely-related species of monkeyflower (Mimulus) -- one
species is pollinated by bumblebees, the other by hummingbirds, and the 12
specific morphological differences that allow this are each traced to one or a
few loci on the chromosomes (Bradshaw et
al., 1998).
-
The only other evidence that Wells cites in favor of his pet theory about
how not just evolutionary biologists but geneticists and developmental
biologists have got things horribly wrong is an anecdote about a single
lecturer he met at a single meeting in Germany in 1999. Based on the vague
description it sounds like the lecturer was critiquing oversimplified models
of connections between genes and development (and environment, which is also
important), but of course anyone in Evo-Devo is well aware of these kinds of
complexities.
-
I know that the tolerant teacher should operate under the philosophy that
there is no such thing as a dumb question, but Wells manages to ask a
fantastically dumb one on page 192. He asks, "If our developmental genes are
similar to those of other animals, why don't we give birth to fruit flies
instead of human beings?" I don't know if Wells thinks that the Intelligent
Designer is continually intervening to keep organisms reproducing after their
kind or what, but several answers that would occur to anyone who has done any
degree of serious thinking on this issue would include (a) similar genes are
not identical genes; (b) small differences in regulation of genes can result
in big changes; (c) the action of a gene (or the protein from a gene) will
depend on both genetic and environmental context; and particularly (d)
different combinations and arrangements of similar genes can result in very
different patterns. Any serious discussion would consider these blindingly
obvious possibilities in detail.
10. Chapter 10: Fossil Horses and Directed Evolution
It is evident from the chapter title that Wells can find no particular beef
with fossil horses; not only do they not make the list of criteria for textbook
evaluation, they don't even make Wells' list of "10 questions to ask your
biology teacher" (at "Ten questions to ask your biology teacher about evolution by Wells").
There is an obvious question ("Aren't fossil horses pretty darn good evidence
for microevolution adding up to macroevolution"), but it seems unlikely that it
will be added to the list. For a detailed introduction to fossil horses see Hunt (1995).
-
What Wells apparently wants people to believe is that the evolution of
horses was directed. Wells argues that biologists have vigorously tried
to correct the old horse icon (linear evolution) because they are all trying
to prove that evolution is undirected. Of course, all anyone can argue
scientifically about such metaphysical matters is that the pattern doesn't
look like it's going anywhere in particular (just like it doesn't
look like the occurence of a tornado is directed in any obvious way).
Whether evolution is directed, or not directed, in some cosmic sense is not
determinable by science. Wells makes this point when he says that the
hypothesis of directed evolution cannot be disproved by the bush-like pattern
of evidence.
-
Wells, though, tries to have it both ways, keeping his pet notion of
directed evolution unfalsifiable by evidence, but also trying to discern
supporting evidence for directed evolution in the fossil horse record.
-
Wells writes (p. 199), "The mere existence of extinct side-branches doesn't
rule out the possibility that the evolution of modern horses was directed. A
cattle drive has a planned destination, even though some steers might stray
along the way." Icons reviewer Jim Dawson called this passage "an
incredible analogy that evokes an image of God as a cowboy" (Dawson,
2001).
-
Wells, in his figure 10-2 (p. 200) on the modern fossil horse record,
appears to be engaging in the incredibly ironic task of trying to restore an
old, discredited icon of evolution! He leaves out most of the extinct side
branches (and there are very many of those in any real treatment of horse
evolution, see Hunt,
1995), and uses an extra-heavy line to trace the lineage from
Hyracotherium to the modern Equus. In the caption he pens the
following earthshaking observation: "Note that although the new pattern is not
linear, it still shows a continuous lineage connecting Hyracotherium
with the modern horse." Of course, there is a similar lineage connecting
Hyracotherium to every extinct species, also, except that they all went
extinct. In fact, (assuming no special creation of species) every currently
living creature will have a continuous lineage back to its ancestors, and
every extinct creature, well, won't. If Wells writes another book on science,
perhaps he'd like to move into physics and discuss the profound truth that you
can't see the sun at night.
-
On page 202 Wells finally gets around to discussing what is probably the
topic that really motivates him (and the ID movement), namely the supposed
nasty philosophical biases inherent in evolutionary biology. This has been
discussed extensively by others elsewhere and has nothing to do with fossil
horses and precious little to do with textbooks, so for the most part we will
move on. Two points should be made about the words like "random" and
"undirected," which Wells digs out of several textbooks (as if the purpose of
a science textbook is to be a philosophical treatise suitable for word-by-word
analysis for conceivably debatable points).
-
Readers should ask themselves: is the weather "random" and "undirected", or
not?
-
If yes, according to Wells you are subscribing to "materialistic
philosophy in the guise of empirical science" and we should expect Wells to
write his next book about the dogmatic materialism of meteorologists (and
statisticians, etc.).
-
If you think that Wells may be overreacting a wee bit to the implications
of such words (perhaps because of the provocation provided by certain
campaigners for atheism such as Dawkins, who is an excellent zoologist but
who should do a better job of keeping his metaphysics separate from his
science), then join the club, and consider the possibility that evolution is
"random" and "undirected" in the very same way that the weather is
considered "random" and "undirected." (for a reasonable discussion of the
philosophical issues and of the motivations of the ID movement see Pennock,
1999)
Along these lines, it is worth pointing out that the first "random and
undirected" quote that Wells brings up (p. 206) is from a textbook by known
Christian Kenneth Miller, a devout Catholic who is the leading biologist in
the anti-ID movement (see his website and
his book, Miller
(1999), which very sympathetically discusses the religious issues
surrounding the modern evolution debate, including the anti-religious
tendencies of several biologists that IDists love to quote).
11. Chapter 11: From Ape to Human: The Ultimate Icon
At long last, we come to The Ultimate Icon, the one on the cover of
Icons of Evolution: the March From Ape to Man. However, at this
point Wells seems to have completely lost track of textbooks, which you may
recall were supposed to be the topic of the book (this Icon does not appear in
Wells' evaluation criteria on page 249, either). The only mentions of textbooks
in this chapter are listed below:
-
(1) Wells says "Most modern biology textbooks do not even mention Piltdown"
(p. 217).
- (2) Apparently the only thing Wells could find that was juicy enough to
quote in a chapter on 'The Ultimate Icon' was from an interview of Stephen J.
Gould in Raven and Johnson's Biology, where Gould gives his well-known
view that "Humans represent just one tiny, largely fortuitous, and
late-arising twig on the enormously arborescent bush of life." Several points
are worth making:
-
-
Interviews are different from the body of the text, and it is precisely
the place where scientists giving their personal views is perfectly
appropriate.
-
Evaluated as science, the only word which is at all questionable in the
Gould quote is "fortuitous." Interpreted charitably it is even more mild
than "random" and "undirected" that were discussed above. Wells of course
packs all his vitriol towards Gould's philosophy of contingency into that
single word, growling in disgust, "Like so many other things we have
encountered, this is not science, but myth." (p. 228)
-
And finally, Gould's statement is in direct conflict with the ape-to-man
icon that Wells purports to be critiquing in this chapter, and which Wells
put on the cover of Icons of Evolution!
-
It seems likely that Wells couldn't find even a single modern textbook that
contained the "Ape-to-Man" icon -- if they did, Wells would be sure to include
them (it is possible than an occasional textbook includes the icon to
criticize it). I myself distinctly remember my general biology professor
putting up such an "Ape-to-Man" icon, and informing the class in no uncertain
terms that this was an inaccurate and outdated view of human evolution. The
professor then put up some of the numerous spoofs and cartoons of the icon
that she had collected, to make the point, and then put up the modern view,
which is that human evolution follows the branching pattern so common
elsewhere in evolution. Unlike in his horse chapter, Wells makes no attempt to
show readers this new, more accurate, icon.
-
For that matter, Wells spends precious little of this chapter discussing
the actual modern fossil evidence. Pages 212-214 are on Darwin's opinions in
Descent of Man, pages 214-216 are about the interpretation of
Neanderthal fossils in the late 1800's, and pages 217-219 are about Piltdown
man, which it seems no creationist book on evolution is complete without; see
the Piltdown Man webpage for extensive references on the topic.
-
Wells actually gets around to discussing an important fossil skull for a
bit on page 219 (1470, a likely Homo habilis, see "Skull KNM-ER 1470"
for a picture and links), but then hares off for six pages interpreting other
peoples' interpretations of the interpretations of anthropologists of the
hominid fossil record. Nowhere does Wells actually give the reader any
specific idea of what the fossils are like, what their measurements and dates
are, etc. Fortunately, an excellent resource on the web allows you to examine
the pictures yourself and draw your own conclusions (The Fossil Hominids FAQ).
-
Wells (p. 220) quotes Henry Gee writing that all the evidence for human
evolution "between about 10 and 5 million years ago [...] can be fitted into a
small box." But appallingly, Wells somehow fails to inform his readers that
plenty of fossils exist from 5 million years ago onward, which, as Eugenie C. Scott
(2001) pointed out in her review of Wells in Science, "is when
humans evolved." Using Wells' standards, a witness at a murder trial would be
permitted to say that he saw nothing untoward between noon and six, and yet
leave out the fact that he saw a murder at seven.
- Wells focuses on controversies surrounding interpretation, but why worry
about that when you can see the fossils for yourself online and make up your
own mind? Go here: "Prominent Hominid Fossils".
12. Conclusion -- Jonathan Wells' book: Science or Myth?
This concludes the tour of Jonathan Wells' Icons of Evolution.
At the end of Icons, Wells includes an Appendix where he "grades" ten
recent biology textbooks, giving most of them an F (Campbell et al.'s
Biology -- probably the most popular college biology text, by the way --
comes out on top with a D+). Wells then includes "warning labels", which look
suspiciously like cigarette warning labels, which Wells thinks should be put in
textbooks.
Has Wells succeeded in making the case which would justify his
harsh judgements? Let us recall what Wells' argument was supposed to be:
"Some biologists are aware of difficulties with a particular icon
because it distorts the evidence in their own field. When they read the
scientific literature in their specialty, they can see that the icon is
misleading or downright false. But they may feel that this is just an isolated
problem, especially when they are assured that Darwin's theory is supported by
overwhelming evidence from other fields. If they believe in the fundamental
correctness of Darwinian evolution, they may set aside their misgivings about
the particular icon they know something about." (Icons, pp.
7-8)
But as we have seen, in every single case, the actual biological experts
in their specific fields of expertise in fact agree that the actual
evidence in their field supports modern evolutionary theory. Furthermore,
many of these scientists have felt sufficiently strongly about this that they
have published critiques of creationist misinterpretations of their work. Many
of these scientists have felt sufficiently victimized by Wells to write specific
rebuttals of him.
Wells might try to argue that he was talking about the "icons" rather than
the general evidence in the field, but still his argument fails. In the cases of
the Miller-Urey experiment, Darwin's tree of life, vertebrate limb homology,
Archaeopteryx, peppered moths, and Darwin's finches, a fair investigation
of the literature has revealed that Wells has no case, and that these "Icons"
are fully deserving of inclusion in biology textbooks. In the cases of the
four-winged fruit fly, fossil horses, and fossil hominids, we discover that
Wells has not even included these cases in his textbook "evaluation" criteria --
perhaps inclusion of these in the criteria would have raised the textbook grades
too much. In any case it is evident that Wells' problems with the four-winged
fruit fly, fossil horses, and fossil hominids are not really with textbooks, but
with extraneous issues -- the real issues in these cases are Wells' bizarre
views about the relationship between genes and development, and his paranoia
that biology textbooks are pushing the view that life is meaningless and
purposeless. I will have a few final words on this subject in a moment, as it is
an oft-recurring theme in antievolutionist writings.
The single "icon" where Wells has some success is with Haeckel's embryos: the
fraction of textbooks that use Haeckel's drawings should replace them with
photographs or more accurate drawings. But even here, the very authority that
Wells cites against Haeckel's embryos, namely M.K. Richardson, has clearly
stated that the actual facts of embryology do indeed support evolutionary
theory, contradicting Wells' interpretation. As Wells' argument is explicitly
based on the views of the experts in their fields, then Wells, to be
self-consistent, would have to concede that he has only scored a point against
certain textbooks, and not against the theory of evolution.
However, let us be generous and grant Wells a full point for the Haeckel's
embryos case. On the other nine "icons," though, Wells has come up empty-handed.
So Wells has earned a 1 out of 10. Even a generous curve would not save Wells
from a flunking grade. One would think that a guy with a Ph.D. from Berkeley
would have done better.
The only thing more discouraging than Wells' grade are the rave reviews that
Wells got from his peers at his current workplace, the Discovery Institute. Wells' fellows at the
Discovery Institute's Center for the Renewal of
Science and Culture lavished praise on Icons of Evolution, with
nary a word of criticism or even mild questioning. Sure, we might expect
antievolutionist demagogues to do their usual braying; for example, in his
typically balanced style Phil Johnson writes of Icons, "This is one of
the most important books ever written about the evolution controversy. It shows
how devotion to the ideology of Darwinism has led to textbooks which are full of
misinformation." But even Michael Behe, who does know some biology and really
should have known better, has apparently become radicalized enough to write,
"Jonathan Wells demonstrates with stunning clarity that the textbook examples
Darwinists themselves chose as the pillars of their theory are all false or
misleading. What does this imply about their scientific standards? Why should
anyone now believe any of their other examples?" This is the same Behe, you will
recall, who accepted the evidence for common descent -- i.e. the tree of life --
as recently as 1996. What is the intellectual status of a movement like
Intelligent Design that cannot bring itself to question even the most outrageous
of Jonathan Wells' distortions? (for the above reviews and others, see
"Reviews of Icons")
I have only had time to refute the major arguments that Wells raises in
Icons; unfortunately this only scratches the surface. A truly thorough
refutation would take a full book, and one rather longer than Icons at
that. I fear that I have not given readers a sufficient impression of just how
deceptive and devious a writer Jonathan Wells is. Through most of the book,
virtually every sentence contains some sort of illegitimate slant,
whether quoting a scientist out of context, or leaving out crucial pieces of
information, or presenting a nonexpert opinion as an authoritative one, or
simply spewing out unsupported exclamations of doubt, derision, and "dogmatic
Darwinism!" Icons is an impressive bit of propaganda, and frankly,
Jonathan Wells is probably the slickest operator that the antievolution movement
has ever produced. His book, packed with quotes and authoritative declarations,
mangling topic after topic in rapid succession, is a calculated attempt to
overwhelm the reader by sheer diversity of material; even the biologically
educated reader is not likely to have the necessary background to spot all of
Wells' tricks. Writing this review required a substantial amount of research and
help from numerous veteran creationism/ID debaters (see Acknowledgements).
But Wells' cleverest move of all was to attack textbooks rather than
taking on the science directly. The all too common response, even from
biologists, has been along the lines of "Well, sure, textbooks have problems,
but this doesn't affect the theory of evolution." This is falling into Wells'
trap. This review has shown that the topics discussed actually do belong in
textbooks, and do constitute good evidence for evolution, according to the
evidence and according to the experts that Wells claims for support. The book
Icons of Evolution is the real scientific travesty.
What is Icons really about?
The central irony of Icons of Evolution is that, while
biologists no longer accept and indeed actively debunk the "March From
Ape-to-Man" image on the cover, it appears to be closer to something that
Wells believes. He apparently does not deny common ancestry of humans
with animals; on page 223 (in the middle of six pages of selective quoting about
the subjectivity and disagreements in paleontology) Wells admits, "Obviously,
the human species has a history. Many fossils have been found that appear to be
genuine, and many of them have some features that are ape-like and some that are
human-like." It seems like Wells' next sentence ought to be "Sorry for all the
trouble, folks, I guess I got a little carried away with this book...", but of
course it isn't. As far as anyone can tell, Wells has the idea that "the human
species was planned before life began, and that the history of life is the
record of how this plan was implemented" (see his essay "Evolution and Design").
In other words, to Wells, evolution (with some unspecified touch of ID)
was marching towards a goal of humans, just like the
Apes-to-Man icon on the cover of Icons of Evolution! It seems
likely that the insertion of this metaphysical idea into science education, as
science, is Wells' real goal.
But is it really necessary to force theology into science? As we saw in the
fossil horses chapter, Wells imputes far more metaphysical significance to words
such as "random" and "undirected" than they actually have scientifically.
Scientifically, evolution is described as "random" and "undirected" in the same
way that the weather, earthquakes, and numerous other natural processes are
described as "random" and "undirected." (For that matter, evolution is also
predictable in a way similar to weather and earthquakes.) Does describing the
weather, or evolution, as somewhat "random" really have the offensive
metaphysical implications that Wells thinks?
For an alternative model, we should investigate the central quote of Wells'
last chapter. Wells is severely offended by Dobzhansky's statement "Nothing in
biology makes sense except in the light of evolution" and decries all of the
evil materialist-naturalist metaphysics he sees in it. But Wells, as usual,
fails to give his reader crucial information: Wells fails to say anything about
Dobzhansky's actual metaphysics: Dobzhansky was a life-long Russian
orthodox Christian. Here are some more quotes from the very same article by
Dobzhansky (1973), which is available online at "Nothing in Biology Makes Sense Except in the Light of Evolution".
"It is wrong to hold creation and evolution as mutual exclusive
alternatives. I am a creationist and an evolutionist. Evolution is
God's, or Nature's, method of creation. Creation is not an event that happened
in 4004 B.C.; it is a process that began some 10 billion years ago and is
still under way."
"Does the evolutionary doctrine clash with religious faith? It does not. It
is a blunder to mistake the Holy Scriptures for elementary textbooks of
astronomy, geology, biology, and anthropology. Only if symbols are construed
to mean what they are not intended to mean can there arise imaginary,
insoluble conflicts. As pointed out above, the blunder leads to blasphemy; the
Creator is accused of systematic deceitfulness." (Dobzhansky T., 1973,
"Nothing in biology makes sense except in the light of evolution", American
Biology Teacher 35:125-9)
13. Acknowledgements
This article was composed with valuable help and comments from Bruce Grant,
Bob Hagen, Wesley Elsberry, Michael Hopkins, Burt Humburg, Ian Musgrave, Pete
Dunkelberg, Jesse, igkappa, theyeti, and several others.
[Talk.Origins
Icons of Evolution FAQs]
14. References
Amundson, R. (2001).
"Homology and Homoplasy: A Philosophical Perspective." Produced by Nature
Publishing Group. Accessed online on 1/15/2002.
Baldauf, S. L., Roger, A.
J., Wenk-Siefert, I. and Doolittle, W. F. (2000). "A kingdom-level phylogeny of
eukaryotes based on combined protein data." Science, 290(5493):
972-977.
Bradshaw, H. D., Otto, K.
G., Frewen, B. E., McKay, J. K. and Schemske, D. W. (1998)."Quantitative trait
loci affecting differences in floral morphology between two species of
monkeyflower (Mimulus)." Genetics, V149(N1): 367-382.
Brandes, J. A., Boctor, N.
Z., Cody, G. D., Cooper, B. A., Hazen, R. M. and Yoder, H. S. J. (1998).
"Abiotic nitrogen reduction on the early Earth." Nature,
395(6700): 365-367.
Campbell, N. A., Reece,
J. B. and Mitchell, L. G. (1999).Biology. Menlo Park, Calif., Benjamin
Cummings.
Cavalier-Smith, T.
(2001). "Obcells as proto-organisms: Membrane heredity, lithophosphorylation,
and the origins of the genetic code, the first cells, and photosynthesis."
Journal of Molecular Evolution, V53(N4-5): 555-595.
Cavalier-Smith, T.
(2002)."The neomuran origin of archaebacteria, the negibacterial root of the
universal tree and bacterial megaclassification." Int J Syst Evol
Microbiol, 52: 7-76.
Chyba, C.(1998)."Origins of life: Buried beginnings."
Nature, 395(6700): 329-330.
Cohen, J. (1995). "Novel center seeks to add spark to
origins of life." Science, 270(5244): 1925-1926.
Collins, A. G. and
Valentine, J. W. (2001)."Defining phyla: evolutionary pathways to metazoan body
plans." Evol Dev, 3(6): 432-442.
Conway Morris, S. (1998). The Crucible of Creation:
The Burgess Shale and the Rise of Animals. New York, Oxford University
Press.
Cook, L. M.(2000). "Changing views on melanic moths."
Biological Journal of the Linnean Society, 69: 431-441.
Copley, J. (2001). "The story of O." Nature,
V410: 892-864.
Davis, W. L. and McKay,
C. P. (1996). "Origins of life: a comparison of theories and application to
Mars." Orig Life Evol Biosph, 26(1): 61-73.
Dawson, J. (2001). "Book review: Icons of Evolution --
Science or Myth?" HMS Beagle(97).
Dobzhansky, T. (1973). "Nothing in biology makes sense except in the light of evolution." American
Biology Teacher, 35: 125-129.
Engel, M. H. and Macko,
S. A. (2001). "The stereochemistry of amino acids in the Murchison meteorite."
Precambrian Research, V106(N1-2): 35-45.
Espiritu, D. J., Watkins,
M., Dia-Monje, V., Cartier, G. E., Cruz, L. J. and Olivera, B. M. (2001).
"Venomous cone snails: molecular phylogeny and the generation of toxin
diversity." Toxicon, 39(12): 1899-1916.
Grant, B. S. (1999). "Fine tuning the peppered moth
paradigm." Evolution, 53(3): 980-984.
Grant, B. S. and
Clarke, C. A. (2000). "Industrial Melanism." Encyclopedia of Life
Sciences.
Grant, B. S., Owen, D. F.
and Clarke, C. A. (1996). "Parallel Rise and Fall of Melanic Peppered Moths in
America and Britain." Journal of Heredity, V87(N5):
351-357.
Grant, B. S., Cook, A. D.,
Clarke, C. A. and Owen, D. F. (1998). "Geographic and temporal variation in the
incidence of melanism in peppered moth populations in America and Britain."
Journal of Heredity, V89: 465-471.
Hunt, K. (1995). "Horse Evolution." Produced by
Talkorigins Archive. Accessed online on 1/15/2002.
Ji, Q., Norell, M. A., Gao, K. Q., Ji, S. A. and Ren, D.
(2001).
"The distribution of integumentary structures in a feathered dinosaur."
Nature, V410(N6832): 1084-1088.
Kasting, J. F. and
Brown, L. L. (1998)."The early atmosphere as a source of biogenic compounds."
in The Molecular Origins of Life: Assembling Pieces of the Puzzle. Andre
Brack. Cambridge, Cambridge University Press: 35-56.
Kelley, D. S., Karson, J.
A., Blackman, D. K., Fruh-Green, G. L., Butterfield, D. A., Lilley, M. D.,
Olson, E. J., Schrenk, M. O., Roe, K. K., Lebon, G. T. and Rivizzigno, P.
(2001). "An off-axis hydrothermal vent field near the Mid-Atlantic Ridge at 30
degrees N." Nature, 412(6843): 145-149.
Knoll, A. H. and
Carroll, S. B. (1999). "Early Animal Evolution: Emerging Views from Comparative
Biology and Geology." Science, 284(5423): 2129 - 2137.
Kral, T. A., Brink, K. M.,
Miller, S. L. and McKay, C. P. (1998). "Hydrogen consumption by methanogens on
the early Earth." Orig Life Evol Biosph, 28(3): 311-319.
Majerus, M. E. N. (1998). Melanism: evolution in
action. Oxford ; New York, Oxford University Press, pp. xiii, 338.
Majerus, M. E. N. (1999). "Personal communication to
Donald Frack." Produced by Calvin College. Accessed online on 1/15/2002.
Majerus, M. E. N. (2000). "A bird's eye view of the
peppered moth." Journal of Evolutionary Biology, 13:
155-159.
Miller, K. R. (1999). Finding Darwin's God: a
scientist's search for common ground between God and evolution. New York,
Cliff Street Books, pp. xiii, 338.
Monmonier, M. (1996). How
to Lie with Maps. Chicago, University of Chicago Press, pp. 1-207.
Murakami, T., Utsinomiya,
S., Imazu, Y. and Prasad, N. (2001)."Direct evidence of late Archean to early
Proterozoic anoxic atmosphere from a product of 2.5 Ga old weathering." Earth
Planet. Sci. Lett., 184(2): 523-528.
NOAA (2001). "What is an El Niño?" Produced by NOAA.
Accessed online on 1/15/2002.
Pennock, R. T. (1999). Tower of Babel: the evidence
against the new creationism. Cambridge, Mass., MIT Press, pp. xviii, 429.
Perkins, S. (2001). "New type of hydrothermal vent looms
large." Science News, 160(2): 21.
Peterson, K. J.
and Eernisse, D. J. (2001). "Animal phylogeny and the ancestry of bilaterians:
inferences from morphology and 18S rDNA gene sequences." Evol Dev,
3(3): 170-205.
Raff, R. A. (2001). "The creationist abuse of evo-devo."
Evol Dev, 3(6): 373-374.
Rasmussen, B. and
Buick, R. (1999). "Redox state of the Archean atmosphere: Evidence from detrital
heavy minerals in ca. 3250-2750 Ma sandstones from the Pilbara Craton,
Australia." Geology, 27(2): 115-118.
Richardson, M. K. (1998).
"Letter." Science, 280(5366): 983.
Richardson, M. K.,
Hanken, J., Gooneratne, M. L., Pieau, C., Raynaud, A., Selwood, L. and Wright,
G. M. (1997). "There is no highly conserved embryonic stage in the vertebrates:
implications for current theories of evolution and development." Anat
Embryol, 196: 91-106.
Sato, A., Tichy, H.,
O'HUigin, C., Grant, P. R., Grant, B. R. and Klein, J. (2001). "On the origin of
Darwin's finches." Molecular Biology and Evolution, 18(3):
299-311.
Scott, E. C. (2001). "Icons of evolution - Science or
myth? Why much of what we teach about evolution is wrong, (2000) (English) by J.
Wells." Science, V292(N5525): 2257-2258.
Segre, D., Ben-Eli, D.,
Deamer, D. W. and D., L. (2001). "The lipid world." Orig Life Evol
Biosph, 31(1-2): 119-145.
Theobald, D. (2002a). "29
Evidences for Macroevolution." Produced by TalkOrigins. Accessed online
on 1/20/2002.
Theobald, D. (2002b). "29
Evidences for Macroevolution: A Response to Ashby Camp's "Critique"." Produced by TalkOrigins.
UCMP (2000). "Metazoa: Fossil Record -- Chart of First
Appearances of Metazoans." Produced by University of California Museum of
Paleontology. Accessed online on 10/10/2001.
Von Damm, K. L. (2001). "Lost City found." Nature,
412(6843): 127-128.
Walsh, C. (2000). "Molecular mechanisms that confer
antibacterial drug resistance." Nature, V406(N6797): 775-781.
Weiner, J. (1994). The beak of the finch: a story of
evolution in our time. New York, Knopf: Distributed by Random House, pp. x,
332.
Zimmer, C. (2001). Evolution: The Triumph of an
Idea. New York, Harper Collins.
15. Endnotes
[1] Nic Tamzek is the pen name of Nicholas Matzke.
[2] The introduction to the book, containing a complete list
of Wells' "icons", can be found online
[3] It may be objected here that Sargent (of the paper
Sargent, Millar, and Lambert, 1998, "The "classical" explanation of industrial
melanism: assessing the evidence," in Evolutionary Biology,
30:299-322), is a moth researcher. This is true, but most of Sargent's
work appears to be on moth species other than peppered moths, such as the
genus Catocala. In genera such as Catocala the situation regarding
industrial melanism and selection appears to be different. Majerus barely cites
Sargent at all in his detailed review of peppered moths in Industrial
Melanism: Evolution in Action.
Regarding the Sargent et al. paper, which is the major source for
those proclaiming the downfall of the classical peppered moth story, an
under-realized fact is that it is formally a review of industrial melanism in
moths in general, and in many species other than the peppered moth
(Biston betularia) the evidence is indeed poor that the "classical
explanation" (selective predation against backgrounds changing due to pollution)
applies. Peppered moths of course come up repeatedly as they are the species on
which the most work has been done, and the authors do question the classical
explanation for peppered moths as well. However, the peppered moth experts are
unimpressed, just as they were unimpressed by a 1986 paper by some of the same
authors (Lambert, Millar, and Hughes (1986), "On the classic case of natural
selection", Rivista di Biologia, 79:11-49.). Here is Cook's (2000)
take:
Work by H. B. D. Kettlewell suggested that selective predation was the main
determining factor in B. betularia, and probably in a wide range of other
examples as well. His evidence consisted of surveys which put the correlation
between melanic frequencies and urbanization on a quantitative basis
(Kettlewell, 1958, 1965), demonstration that wild birds would eat the moths if
they found them (Kettlewell, 1955), and the now famous demonstration that
birds discovered most readily the forms least like the daytime backgrounds on
which they rested (Kettlewell, 1973; Rudge, 1999). Colour of resting
background and heterogeneity due to epiphyte cover, appeared to affect
relative visibility. Selective predation became the accepted explanation for
the rise in morph frequency (Majerus, 1998).
Since then, further evidence has been collected. Over the last two decades
industrial environments have become cleaner and melanic frequencies lower
(Clarke et al., 1990; Mani & Majerus, 1993; Grant et al., 1998;
Cook et al., 1999). There has also been some revision of interpretation. It
has been shown that experiments designed to detect and measure selective
predation were carried out in places where moths were not usually likely to
rest if left to their own devices (Mikkola, 1979, 1984; Liebert &
Brakefield, 1987; Grant & Howlett, 1988; Majerus, 1998). There is by no
means a one-to-one relation between reversion of morph frequency and reversion
of epiphyte pattern (Bates et al., 1990; Grant et al., 1998). These findings
cause us to reexamine the story, but they do not obviously require a radical
revision. Estimations of selection appear to show a correspondence between
fitness and frequency. The correlation between the condition of the sites used
in experimental studies and those actually used by the insects is likely to be
high. Further lines of experimentation are suggested, but no previously held
view has been overturned.
The general tone of commentary on Biston studies has, however, altered.
From being treated as a vivid demonstration of natural selection (Luria, Gould
& Singer, 1981, provide an excellent example) and good field
experimentation (Hagen, 1999), the work concerned has come to be viewed with
suspicion (Sermonti & Catastini, 1984; Cherfas, 1987). In a recent review
by Sargent et al. (1998) almost every reference to past work is
predicated by expressions of doubt, reworking ground covered by Lambert et
al. (1986). When discussing predation experiments they conclude ". . .
there seems to be no clear and consistent relationship between the relative
survivorship of different morphs . . . and the frequencies at which the morphs
naturally occur in different environments". Coyne (1998) adopts a similar
tone, saying that the flaws in the work are too numerous to list. This has led
to some alarming reporting, such as Matthews (1999) in the Daily Telegraph
newspaper in Britain, who refers to a "series of scientific blunders" and
states that the experiments are "now thought to be worthless". This article in
turn was linked in its electronic web version to the Creation Science home
page. Recent commentaries are quoted on more than one anti-evolution web site.
A balanced account, which shows the strength of the data in the face of recent
criticism, has been provided by Grant (1999). I propose here to illustrate the
predation results, which Sargent et al. did not do when they criticized
them, and to consider why a radical change in view should have
occurred.
Cook's conclusion about the state of the evidence and the somewhat baffling
criticism the peppered moth story has received:
CONCLUSION
In industrial melanism of Biston betularia, both the original
increase and recent decline in frequency of melanics are striking examples of
natural genetic change closely related to change in the environment. They must
have a selective basis. The experiments demonstrate selective removal. There
is a general correspondence of morph frequency and appearance of backgrounds
likely to be adult resting sites. None of this is in doubt. The evidence is,
however, limited in two ways. First, non-visual components of selection have
not been investigated directly in this species. Analysis of segregating
progenies suggests pre-adult survival differences (Creed et al., 1980) with
carbonaria homozygotes having an advantage over other morphs.
Non-visual selection is certainly indicated in studies of other melanic moths
(Bishop & Cook, 1980), but we have little more idea than Leigh (1911) how
it may operate.
Secondly, the experimental and observational evidence cannot on its own
carry the burden of a particular view of evolution, such as found in Oxford
ecological genetics. Smocovitis (1996) describes how the view of the
[Neodarwinian Modern] Synthesis with which it was associated came to seem
'constricted' to many students of evolution, and to generate a reaction in
favour of more complex models; the last three decades have been a period of
lively debate and controversy. Distrust of the evidence of industrial melanism
may sometimes arise from a wish to question how the example relates to more
complex levels of evolutionary theory. Criticism on these grounds is
misplaced, and can attract the attention of advocates of creationism who see
an evolutionary field in apparent disarray. The Biston story continues
to provide an exceptional opportunity to analyse a pattern of selection. It
should be pursued, along with study of other species with related but
different responses to environmental change.
It should also be pointed out that while Sargent appears skeptical of some of
the evidence for the classical peppered moth story, he has no problem with the
general idea of cryptic (camouflage) coloration in moths; he writes that the
classical explanation is "eminently reasonable", and furthermore he has himself
published on crypsis in moths. A very interesting webpage on Sargent's
observation of the behavior of another moth species, Catocala relicta, is
online.
Notably, the evidence is against this kind of "find a matching
background" behavior in peppered moths. Peppered moths appear to just look for
shaded spots (such as beneath trunk-branch joints), whether or not the moth is
dark or light colored. As Sargent et al. grudgingly acknowledge, this is
actually a good thing for the classic selection-by-bird predation story for
peppered moths, as picking-matching-background behavior would actually
constitute negative feedback that would weaken the effects of natural
selection (i.e., as tree surfaces got darker, the moths would just behaviorally
move to lighter surfaces that matched their camouflage).
Regarding Jerry Coyne's (1998) review of Majerus' book Industrial
Melanism in Nature,
readers should not allow Coyne's excellent reputation as a
nonsense debunker to dissuade them from recognizing that Coyne's review contains
at least one glaring mistake: namely, Coyne writes, "Majerus notes that the most
serious problem is that B. betularia probably does not rest on tree
trunks -- exactly two moths have been seen in such a position in more than 40
years of intensive search. The natural resting spots are, in fact, a mystery."
However, Coyne somehow fails to mention that in the very next paragraph of
Majerus' book (pp. 121-122), Majerus cites his own data on the natural resting
places of moths -- some 47 moths -- not a lot, but far more than two. This data,
listed in Table 6.1 of Majerus' page 123, indicates that peppered moths in fact
rest in diverse locations (12 on tree trunks, 20 on "trunk-branch joints", and
15 in tree branches). Similar percentages are found in Table 6.2 on a larger
dataset of resting moths found near traps.
All of this serves to illustrate that Coyne's opinion on the classical
peppered moth explanation is by no means definitive. Wells, of course, gives
readers very little of the above complexity of the true situation, and instead
quotes the juicy bits of Coyne's review. Wells' own treatment of moth resting
locations appears to be deliberately deceptive. Wells refers to Clarke et
al's (1985) reference to the two moths collected (Clarke was basis for
Coyne's remark also), and goes on to selectively quote two additional papers on
the topic of resting locations, as well as Majerus' book, to support the notion
that moths don't rest on tree trunks. In the notes (p. 304) Wells buries a
reference to Majerus' book amongst the other papers, and refers readers only to
page 116 of Majerus' book, when in fact the section "The resting behaviour of
peppered moths", including the tables, is found on pp. 121-123. Why does Wells
not just put the numbers, which are the best anyone has, in the text for his
audience to read?
This essay first appeared at TalkOrigins.Org
* * *
Location of this article:
http://www.talkreason.org/articles/icon.cfm